THIS CONTENT IS BROUGHT TO YOU BY NTNU Norwegian University of Science and Technology - read more
New therapy can treat rare and hereditary diseases
Researchers are on the hunt for treatments for diseases that have been deemed incurable.
Diseases that are widespread in large parts of the population have been extensively researched for many decades. This includes diseases such as cancer and heart diseases.
As a result, treatment methods have improved enormously thanks to long-term research efforts.
However, there are many diseases that affect just a handful people. These diseases often fly under the radar and are far less studied. They include quite a few rare, hereditary diseases.
A team of researchers is now in the process of trying to change this.
“For some hereditary, rare diseases, there is currently no cure. However, gene therapy is a possible solution, and we are now testing various strategies using gene therapy,” Magnar Bjørås says.
He is a professor at NTNU’s Department of Clinical and Molecular Medicine. He has established a research team at NTNU and Oslo University Hospital. They conduct basic research on rare hereditary diseases. Their long-term goal is to find new gene therapies.
Brain cells become dysfunctional or die
One of the rare diseases for which there are currently no medications or treatments available is called DOOR syndrome.
This is a congenital disorder that involves multiple abnormalities. DOOR is an acronym for the main features of the disorder: Deafness, Onychodystrophy (short or absent nails), Osteodystrophy (short fingers and toes) and developmental delay and intellectual disability (previously called mental Retardation).
DOOR syndrome is hereditary and is caused by the lack of a specific protein in the genes called OXR1 (OXidation Resistance gene 1).

“Due to lack of this protein, the brain cells are unable to develop as they should. As a result, the brain cells either become dysfunctional or simply die,” Bjørås says.
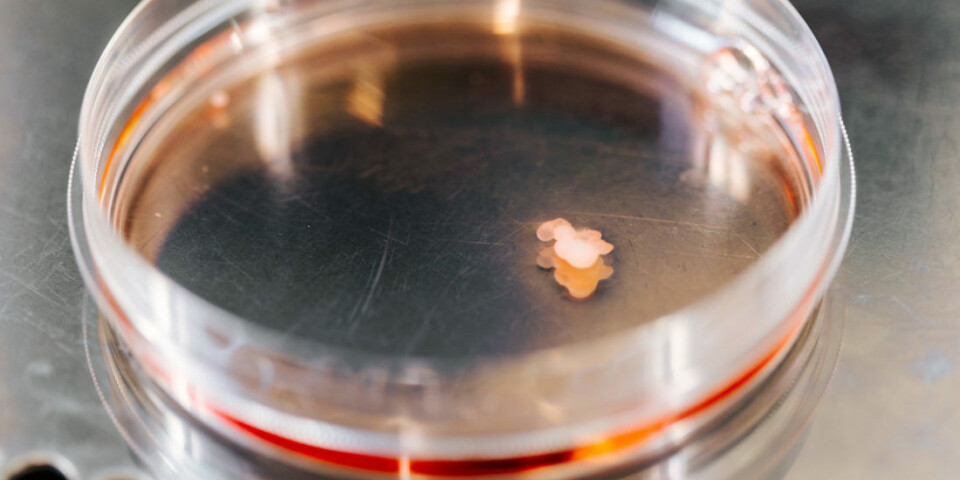
In order to investigate whether there is a way to prevent this from happening, the Bjørås team has carried out tests inside mini-brains that they grow in their lab.
From skin cells to mini-organs
The Bjørås team has been working on growing mini-organs such as mini-brains, mini-lungs, and mini-eyes since 2018. The researchers use the mini-organs to test drugs and gene therapy.
In order to grow mini-brains for their research on DOOR syndrome, the research team needed cells from people who have this disease.
A number of cases have been registered in Canada and the Middle East, and the research being done in Norway is based on skin cells from people who have DOOR syndrome.
“In the laboratory, we have transformed the skin cells into embryo cells. We have reversed the development in the skin cells so that they return to the foetal stage and become like the first cells that form in humans. We have then used these stem cells to create mini-brains,” Bjørås says.
Using skin cells from people with DOOR syndrome, the researchers have recreated the disease in the mini-brains. They can then use these mini-brains to test out therapies for this disease.
The process of developing mini-brains takes several months and is painstaking and expensive.
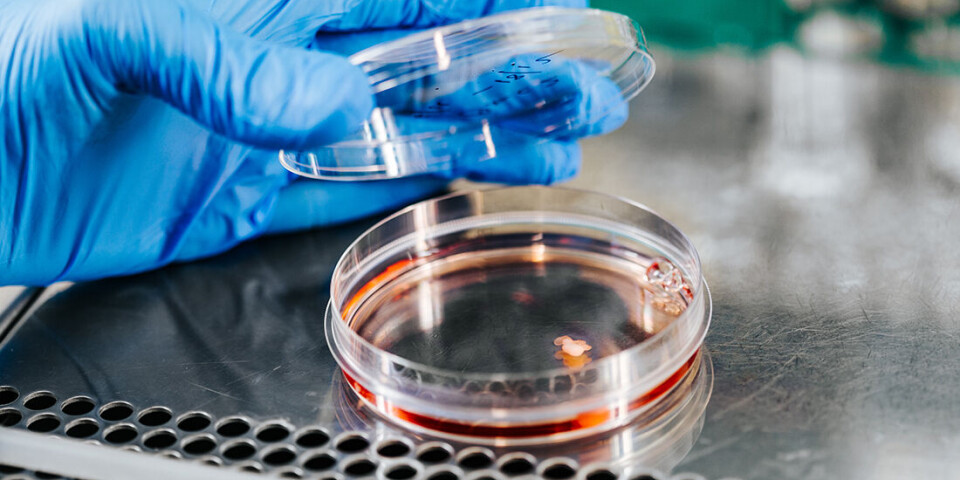
Starting production of the missing protein
The work has given the researchers insight into the reasons why patients develop disease – and thus also ideas for treatment strategies.
Gene therapy is one possible treatment where the brain cells can be made to start producing the missing protein.
A virus is actually used as a messenger that delivers the necessary production information to the brain cells.
“As a follow-up to our published work, we are now testing virus-based gene therapy as a treatment for this disease,” Bjørås says. “We create a harmless virus in the lab and then put a healthy OXR1 gene into the virus’ genome, and this gene has the ability to produce the protein that brain cells lack in people with DOOR syndrome.”
Can stop the disease
The virus is then injected into the mini-brains.
“The virus is absorbed into the brain and brain cells. The gene introduced into the brain cells via the virus can then begin to produce the missing protein,” Bjørås says.
If this protein can be overproduced, it helps to stop and, at best, reverse the disease. In order to treat DOOR syndrome, patients will need to start gene therapy at a very early stage, probably as soon as the first symptoms of disease are noticed.
Gene therapy research has evolved greatly over the past 20 years.
“In 2007, there was only one clinical trial of gene therapy. Today, there are thousands of clinical trials involving gene therapy,” Bjørås says.
Leading the way in treating other hereditary diseases
The research has not only provided new knowledge and ideas for the treatment of patients with DOOR syndrome, but also for other diseases.
“What is particularly interesting about the OXR1 protein that patients with DOOR syndrome lack is that this gene therapy method also has an interesting potential for treating other diseases,” Bjørås says.
The OXR1 protein reduces inflammation, which is a characteristic of most degenerative diseases of the brain, such as childhood dementia, Alzheimer’s, ALS, and Parkinson’s.
Studying brain development
The researchers have used new, advanced technology for this research. The mini-brains that they have grown are made from skin cells from DOOR syndrome patients and from healthy individuals.
The mini-brains have been used to study brain development and the scientists have created different parts of the brain that control different functions, such as memory, learning, motor skills, fluid balance, hormone balance, and temperature control.
It has been shown for the first time in a human model that OXR1 promotes protein methylation protein methylation during brain development in space and time. Protein methylation is one of several important chemical processes that control gene expression.
The study provides new insights into pathological traits associated with OXR1 deficiency in patients.
Reference:
Lin et al. A loss-of-function mutation in human Oxidation Resistance 1 disrupts the spatial–temporal regulation of histone arginine methylation in neurodevelopment, Genome Biology, vol. 24, 2023. DOI: 10.1186/s13059-023-03037-1
More content from NTNU:
-
Study: Many teens sext on social media
-
Are boys more motivated and girls more compassionate?
-
Why seafarers have little confidence in autonomous ships
-
These house sparrows can help us save other endangered animals
-
Experienced therapists can give psychology students an extra boost
-
Teamwork is a skill – it must be practiced





































