This article is produced and financed by NTNU Norwegian University of Science and Technology - read more
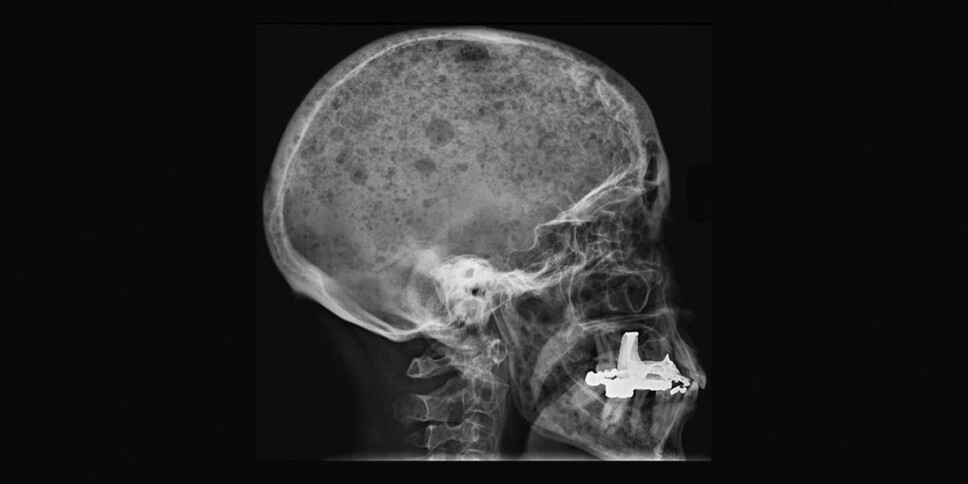
Bone marrow cancer: Perforated bone tissue from too little sugar
Bone marrow cancer is currently an incurable disease that affects about 400 people in Norway every year. Professor Therese Standal at NTNU has now found an important reason for bone destruction in people with this disease.
Could something as simple as a certain type of sugar water be medicine for perforated bones, and even bone marrow cancer itself?
Inside our bodies are some jellyfish-like cells that actually eat away at our bones. Every year, they eat about ten per cent of the bone mass in our body. Fortunately, other cells usually follow and build up new bone.
We undergo a kind of continuous remodelling and repair that enables most of us to traipse around with steel in our legs and arms.

Bone-eating cells run amok
In people with bone marrow cancer, the bone-eating cells run amok. They become too numerous and eat too much. The bone-building gang doesn’t have time to rebuild the bone mass, despite overtime and long shifts. Bone tissue gets gobbled up.
Many people with bone marrow cancer often end up with perforated bones, a condition that is very painful to live with. They sometimes experience collapsed vertebrae or suffer broken bones just by turning in bed.
For decades, scientists around the world have been scratching their heads and wondering what the cause could be. Various theories have been launched, but researchers have not reached a unified theory on what the main cause is.
Too many unusable antibodies produced
Bone marrow cancer remains an incurable disease so far. The available treatment can prolong life, but not cure the disease.
Now Standal and her research group at CEMIR at NTNU have discovered a piece of the puzzle that looks very promising.
They have come to the conclusion that the cause of the bone destruction is too little sugar. We’re not talking about the sugar we eat in our cakes and biscuits, but sugar that resides on a substance that is important for the immune system.
To get to the bottom of how sugar is related to bone loss, we need to get into the bone marrow. This is the soft cavity inside all our bones.
Within the bones are plasma cells. When bacteria or viruses enter the body, the plasma cells begin their job of getting rid of the invaders. Antibodies are produced which are sent via the blood, ready to do battle.
So far so good, but in people with bone marrow cancer, far too much of one type of antibody is produced. It’s going amok here, too. The antibody that the cancer makes is also completely useless. It doesn’t knock out either the cold or the flu but just takes up too much space and displaces other types of antibodies.
Search for an answer took five years
“I thought simply. If people with bone marrow cancer have too much of the antibody and too many bone-eating cells, then they must be connected,” Standal says.
The search for an answer gobbled a lot of her working hours for almost five years. The hard work was fortunately not in vain, and has led to a completely new and fundamental understanding.
The finding has now been published in the highest ranked blood disease journal in in the world.
This is how Standal arrived at the answer:
The vast majority of patients with bone marrow cancer develop perforated bones, but not all. Standal asked nicely, and received samples from patients with bone loss. She also took samples from patients without this kind of bone loss.
The researchers extracted antibodies from the samples and cultured bone-eating cells in the laboratory.
- When Standal placed the bone-eating cells into the antibody of the patients with bone perforations, she discovered that the number of bone-eating cells increased.
- When she put the bone-eating cells into the antibody of the patients without bone perforations, she discovered that the number of bone-eating cells did not increase.
Removed and added sugar
“Why that was the case became the next interesting thing to figure out,” Standal says.
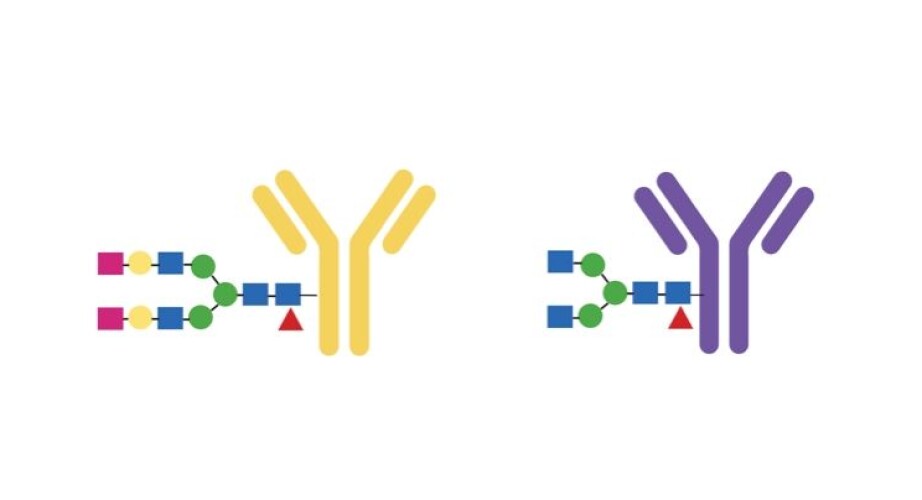
The antibody carries a type of sugar that “decorates” it, in a way. The sugar has an effect on how the antibody works. Standal found her way to Manfred Wuhrer at the Center for Proteomics and Metabolomics of the Leiden University Medical Center in the Netherlands. He is a specialist in this type of sugar, and Standal sent the samples to him.
He found that individuals with bone loss were missing two sugar molecules at the end of a long chain inside the antibody.
“There was too little sugar,” says Standal.
But this answer wasn’t sufficient, either.
Although a difference was detected between the two groups, the researchers could not confirm that the missing sugar molecules were the reason patients developed more bone-eating cells. Several further experiments had to be conducted.
The research team went to the lab and put more sugar on the antibody. This did not lead to more bone-eating cells. Standal also did the opposite, removing sugar from the antibody. This did lead to more bone-eating cells.
Fed mice sugar water
The researchers then had sufficient test results to show that too little sugar can be decisive for the number of bone-eating cells. But this is not enough in medical research – at least not if the goal is to use the knowledge to make medicine for humans.
The next step involved animal experiments with mice that have bone marrow cancer. The mice were divided into two groups and were given two different types of sugar water. In theory, one type of sugar water would lead to more sugar on the antibody.
“The theory actually worked. The mice that received this type of sugar water had smaller perforations in their bone tissue. They also developed less cancer,” says Standal.
Now she has to carry out more animal experiments to move forward on the path towards a treatment that can give patients with bone marrow cancer a better life.
“I think it might be realistic to try this on a small group of patients in four to five years,” says Standal.
Reference:
Marita Westhrin et.al.: Monoclonal immunoglobulins promote bone loss in multiple myeloma. Blood, 2020. Summary https://doi.org/10.1182/blood.2020006045
———
Read the Norwegian version of this article at forskning.no


































